 Analysis of FDA Food Registration Compliance Points and Cost Evaluation for Small Volume Trial Orders Exported to the U.S.
Analysis of FDA Food Registration Compliance Points and Cost Evaluation for Small Volume Trial Orders Exported to the U.S.introductory
Research shows that 93% people suffer from the "illusion of knowledge" - thinking they know, but actually knowing nothing. Many Taiwanese food producers are preparing toSmall exportsIn the U.S. market, there is often a misunderstanding that "registration is not required if the number of trial orders is small", and this perception gap often leads to the goods being stuck in the customs, delayed customs clearance, or even forced to destroy the painful experience. In this article, we will analyze the situation from a practical point of view:Do you need FDA food registration for small volume export?,green circle(How cross-border compliance platforms, such as Greenoo, can assist you in completing your registration, and how to proceed.FDA Food Registration Cost AssessmentWe are committed to making sure that you are well prepared before you enter the U.S. market.
"This article will focus on the details of FDA registration fees. If you have not yet completed basic registration or are unsure of the overall process, we recommend reading our general overview first:[Taiwan Food Exports to the United States: Complete Guide to FDA Registration (2026)], establishing a global view."
Summary of Core Points
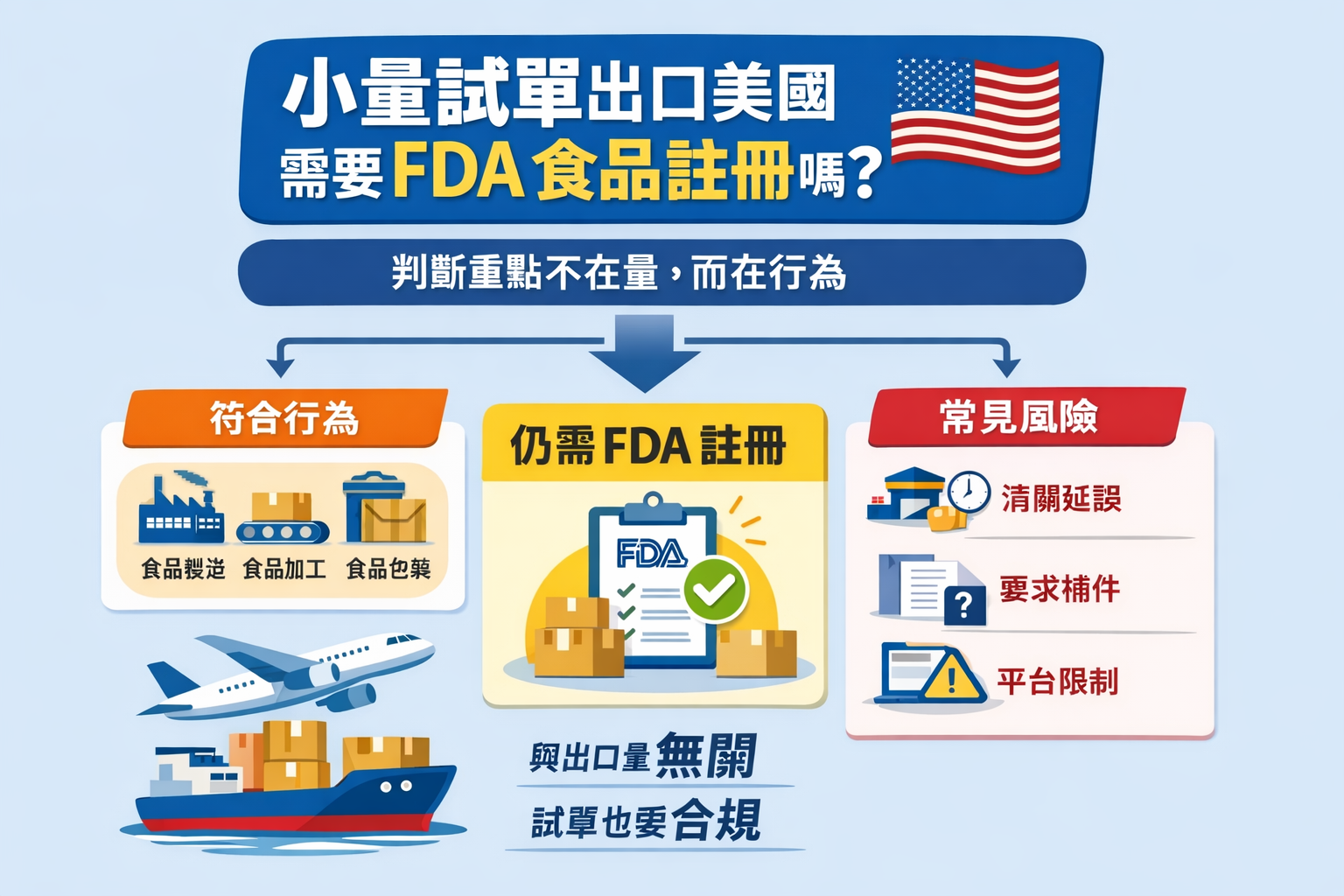
- Regardless of the quantity exported, as long as the food enters the U.S. market, in principle, it is required to complete the FDA food facility registration.
- Trial sales or small orders do not constitute a reason for exemption. Failure to register exposes you to the risk of delayed customs clearance, fines and even return shipment.
- The responsibilities of the factory and the brand need to be clarified, usually the actual manufacturer is responsible for registration, but the brand also needs to ensure compliance.
- Registration costs include initial registration fee, agency service fee and subsequent annual renewal fees, which need to be planned in advance.
- Professional platforms such as Green Circle can provide one-stop compliance services, greatly simplifying the registration process and reducing risks.

FDA Food Facility Registration Process and Common Compliance Blind Spots for Small Volume Exporters
I. Do I need a full FDA registration for a small trial order? Analysis of Regulatory Practices
1.1 Legal Basis for FDA Food Facility Registration
According toU.S. Food and Drug Administration (FDA)Under the Bioterrorism Act of 2002 (Bioterrorism Act) and its successor, the Food Safety Modernization Act (FSMA).Any facility that manufactures, processes, packages, or stores food for consumption in the United States must register with the FDA.The requirement does not set a "quantity threshold" or a "trial sale exemption". There is no "volume threshold" or "trial sale exemption" in this requirement.
important reminderFDA compliance is based on "behavior" not "quantity". Whenever your product enters U.S. commerce, whether it is a sample case or a full container, it is subject to the same regulatory requirements.
1.2 Common Misconceptions: The Risks of "Small Volume Registration"
Many first-time exporters believe that:
- "I'm just trying out a few cases. I don't think I need to register, do I?"
- "It's not too late to register when you know there's a market."
- "I tested it on Amazon FBA in small quantities, and the platform didn't ask for registration."
According toU.S. Customs and Border Protection (CBP)the practice of law enforcement.Food shipments that have not completed FDA registration will be marked as Refused Entry upon entry.It is possible to face:
| Risk Type | Actual Consequences | Financial Impact |
|---|---|---|
| Customs clearance delays | 7-30 days of cargo being stranded in port | Warehouse fees accumulate daily |
| Mandatory Inspection | FDA imposes hold for inspection | Inspection Fee $258-$301/hour |
| Return or Destruction | Inadmissible goods | Loss of full payment + logistics fee |
| Platforms off the shelves | Amazon and other platforms stop selling products | Sales Interruption + Account Risk |
1.3 Do you need FDA food registration for small volume export?What is the answer? Authoritative Answers
The answer is yes.. Whatever you are:
- Trial order of 5 boxes of tea leaves to test the market response
- Selling 50 bottles of jam through e-commerce platforms
- Exhibit with 100 packs of snacks for marketing
FDA Food Facility Registration is required whenever a product enters the U.S. for human consumption.This is a "ticket" to enter the U.S. market, not a "post-scale requirement. This is a "ticket" to enter the U.S. market, not a "requirement after scaling up".
Registration Responsibility: Factory vs.
2.1 Who should be responsible for registration?
According toFDA Food Facility Registration Guide::
Manufacturer Registration Responsibilities::
- A factory that actually produces food must be registered as a "food facility".
- Obtain a unique FDA registration number (11 digits)
- Updated every two years (October through December of even-numbered years)
Brand owners' responsibility::
- Confirmation that the partner factory has completed FDA registration.
- Provide the correct factory registration number at the time of importation.
- For Private Label, make sure the labeling facility is also registered.
2.2 Role of Traders and Agents
Important Concepts: Pure traders or export agents are usuallyunnecessaryregistered as a food facility because they do not actually "manufacture, process, package, or store" food. But they need to:
- Ensure registration of supplier factories is completed
- Fill out the FDA Prior Notice correctly (Prior Notice)
- Designation of U.S. Agent (US Agent)
Three,green circleHow can professional platforms such as this one assist in compliance?
3.1 green circle(Greenoo) Service Overview
green circleIt is a compliant service platform focusing on cross-border e-commerce in Taiwan, Hong Kong and Macao:
- One-stop FDA registration serviceIncludes factory registration, assignment of U.S. agent, and preparation of documents.
- Instant Compliance Consultation: Answer practical questions about small-volume exports, trial orders, etc.
- Cost transparency: Evaluate registration fees and timeline in advance
- Multilingual Support: Traditional Chinese language service to reduce communication barriers.
According toRegistrar Corpand other professional and regulatory organizations.Reduce 40-60% of time cost for FDA registration through professional platform.and significantly reduces the risk of returns due to documentation errors.
3.2 DIY vs Professional Service Comparison
| Project | Self-administration | Throughgreen circleand other platforms |
|---|---|---|
| Time Costs | Requires 3-5 days to study the statute | 1-2 days to complete |
| Preparation of Papers | Easy to miss key information | Specialized assistance in checking |
| U.S. Agents | You need to find it by yourself | Platforms are provided directly |
| Reminder of future updates | Self-recorded | Automatic Notification |
| Fee | Surface free (high time cost) | $ $300-$800 (depending on services) |

Complete Cost Structure Analysis and Budgeting Recommendations for FDA Food Registration
Four,FDA Food Registration Cost Assessment: Complete Budget Guide
4.1 Direct cost items
1. Official FDA Fees
- Registration of Food Facilities: Currently free (FDA direct enrollment system)
- Re-check Fee: $$301/hour for foreign facilities and $$258/hour for local facilities if re-inspection is required for violations (based onFDA Fiscal Year 2020 Announcement)
2. U.S. Agent Service Fee
- Annual fee range: $$100-$$300
- Responsibilities: Act as a bridge between the FDA and the factory and assist in handling emergency notifications.
3. Professional agency fees
- Basic Registration Service: $$300-$$500
- Full compliance package (including labeling review, prior notification, etc.): $$800-$1,500
4.2 Hidden Costs and Long-Term Expenses
Time Costs::
- Self-study of FDA regulations: 15-20 hours
- Document preparation and communication: 8-12 hours
- If the hourly rate is $$30, the time cost is about $$690-$960.
Risk Costs::
- Delay in customs clearance due to non-registration: Warehouse fee $50-$150 USD/day
- Return of goods: return logistics fee + loss of value of goods
- Product Take Down: Sales Disruption + Loss of Goodwill
Annual maintenance costs::
- Renewal of registration every two years: $$100-$$200
- Consultation on Continuing Compliance: $$200-$$500/year
4.3 Proposals for cost-benefit analysis
Expert Advice: For annual exports of **$$10,000 or moreThe ROI (Return on Investment) for investing in professional compliance services is typically in the range of $4,000 to $4,000 per year.300% or above**, as it can avoid major losses such as customs clearance delays and penalties.
According toMcKinseyAccording to a cross-border e-commerce research report, **compliance costs should account for 3-5%** of the CIF cost of a product to be considered a reasonable allocation. For small trial orders, it is recommended that:
- First Trial Order ($500-$1000): Invest at least $$500 to complete basic registration
- Scale-up phase ($5,000+ monthly sales): Consideration of Annual Compliance Program ($$1200-$$2000)
- Multi-Category Expansion: Additional $$300-$$500 compliance budget for each new product line added
V. Practical Operation Steps: Complete Compliance Path for Small Volume Exports
5.1 Pre-registration Preparation (30 days before shipment)
Step 1: Identify Product Categories
- Check if the product is an FDA regulated food.
- Confirmation of the need for additional certification (e.g., low-acid canned food, infant formula)
Step 2: Organize Factory Information
- Factory Name (Chinese and English)
- Full address
- Contact Information
- Product Category Description
Step 3: Select Service Methods
- Evaluation of self-organization vs.green circleand other platforms
- Budgeting and Timeline Planning
5.2 Registration and Execution (to be completed 15 days prior to shipment)
Step 4: Submit for FDA Registration
- ThroughFDA FURLS SystemOnline Registration
- or commissioning an agency service
Step 5: Obtain a Registration Number
- FDA Issues 11-Digit Registration Number
- Maintaining the Registration Confirmation Letter
Step 6: Appointment of U.S. Agent
- Signature of Agent Agreement
- Confirm that the agent information has been correctly entered into the FDA system.
5.3 Final inspection before shipment
Step 7: Prepare Prior Notice
- Submit 2 hours to 5 days prior to shipment
- Must contain FDA registration number
Step 8: Verify Business Documents
- Commercial Invoice with factory name and FDA registration number.
- Packing List
- Bill of Lading
Step 9: Confirmation with Logistics Provider
- Ensure customs brokers are aware that the product is an FDA-regulated food.
- Provide a complete list of compliance documents
Common Violation Cases and Prevention Strategies
6.1 Real Case Alert
Case 1: Tea Trial Order Detained
A Taiwan tea trader exported 30 kilograms of high tea to the U.S. for the first time. Due to the lack of FDA registration, the goods were detained at the port of Los Angeles for 21 days, incurring a storage fee of US$$1,260, and were released only after the registration was completed, resulting in a total loss of more than US$$2,000.
Case 2: Jam brand taken off Amazon shelves
A Taiwanese fruit sauce brand through the FBA trial sale of 50 cans of products, sales of a month after the consumer reported that does not show the FDA registration information, the product was taken down by the platform, the health of the account was seriously damaged.
Case 3: Repeated violations with high fines
A food factory was seized again on the second export after failing to complete improvements after the first violation. FDA demanded a re-inspection fee of $$301/hour x 8 hours = $$2,408 and faced an Import Alert (I.A.).
6.2 List of Preventive Measures
✓ Initiate the registration process 30 days before shipment
✓ Create backups of compliance files (electronic + paper)
✓ Set up a reminder to update in October-December of even-numbered years.
✓ Establishment of Standard Operating Procedures (SOPs) with logistics providers
✓ Regularly review FDA regulatory updates (subscribe)FDA Official E-Newsletter)
VII. Latest Regulatory Trends and Future Outlook for 2025
7.1 FSMA Enforcement Enhancement
According toU.S. Food Safety News (Food Safety News)reported that the FDA in 2024-2025 will beStrengthening Inspection of Small ImportersThis is a special focus:
- Cross-border e-commerce platforms (Amazon, eBay, etc.)
- Direct-to-Consumer (D2C)
- Social Media Sales Pipeline
7.2 Technical Tool Development
The Rise of Digital Compliance Tools::
- Blockchain Supply Chain Traceability System
- AI-assisted label review tool
- Real-time compliance monitoring platform
green circleplatforms are actively integrating these technologies to provide more convenient solutions for small volume exporters.
7.3 Cost Trend Forecast
According toDeloitteThe industry analysis report:
- Compliance Service Fees Projected to Rise 15-20% in 2025-2027(due to labor and system maintenance costs)
- However, the spread of automated tools will reduce the cost of 30% time.
- Overall compliance cost ratio is expected to decrease from 5% to 3%.
Conclusion: A small investment that significantly reduces risk.
Do you need FDA food registration for small volume export?? The answer is very clear--Absolutely.This is not an "option" but a "basic threshold" for entry into the U.S. market. This is not an "option" but a "basic threshold" for entering the U.S. market. Many companies have underestimated the importance of compliance, resulting in failed trial orders, missed market opportunities, and even the negotiation of subsequent large orders.
Key Recommendations for Action::
- Evaluate Now: Review your current export program to confirm that FDA registration has been completed.
- Professional Consultation: Contactgreen circleCustomize your product by using a compliant platform, such as theFDA Food Registration Cost Assessment
- Budget Planning: Incorporate compliance costs into product pricing and market testing budgets.
- Establishment of SOPs: Develop standardized compliance processes to avoid repetitive errors
Final ReminderInstead of facing the anxiety of having your goods detained at customs, invest $500-800 USD now to complete your professional registration. This cost in exchange for a smooth customs clearance experience, stable sales channels, and long-term brand reputation.Compliance is not a cost, it is a strategic investment in the U.S. market.
Do it now.: Interviewsgreen circleGet a free compliance assessment and get your path to the U.S. market on a solid footing from the very first step!
Frequently Asked Questions FAQ
FAQ 1
Q: Do I need FDA food registration for exporting small quantity trial order to USA?
A: The need for FDA food registration depends on whether or not the act of manufacturing, processing, packaging, or storing the food is involved, not on the quantity exported. Even if a small quantity is exported on a trial basis, as long as it meets the definition of a food facility, it is still required in principle to complete an FDA food registration.
FAQ 2
Q: Are there any exemptions from FDA food registration for small quantity test order exports?
A: Currently, FDA regulations do not provide a general exemption from food registration for trial orders or small exports. Whether or not registration is required depends on the role of the actual food facility and the behavior of the operation.
FAQ 3
Q: What are the risks of exporting small quantities without completing FDA food registration?
A: In practice, there may be delays in customs clearance, being asked to submit additional FDA food registration information, or being asked to submit additional documents during e-commerce platform audits, which in turn affects shipment and sales schedules.
FAQ 4
Q: How can Taiwanese sellers determine if they need FDA food registration?
A: It is recommended that the determination be made from three perspectives, including whether it is a food product, whether it involves a food device such as manufacturing or packaging, and whether the product enters into circulation in the U.S. market, rather than based solely on the number of exports.
FAQ 5
Q: If I have completed the FDA food registration, will I not be inspected again if I export a small amount of food?
A: Completion of an FDA food registration does not necessarily mean that you will not be inspected. In practice, you may still be subject to inspection for sampling, data consistency, or other compliance factors, but completing the registration will help to minimize the associated risks.
Reference Sources::
- U.S. Food and Drug Administration (FDA) Official Website
- U.S. Customs and Border Protection (CBP)
- FDA Food Facility Registration Guide
- Registrar Corp Compliance Study
- McKinsey's Cross-Border E-Commerce Analysis
- Deloitte Industry Trends Report
- U.S. Food Safety News