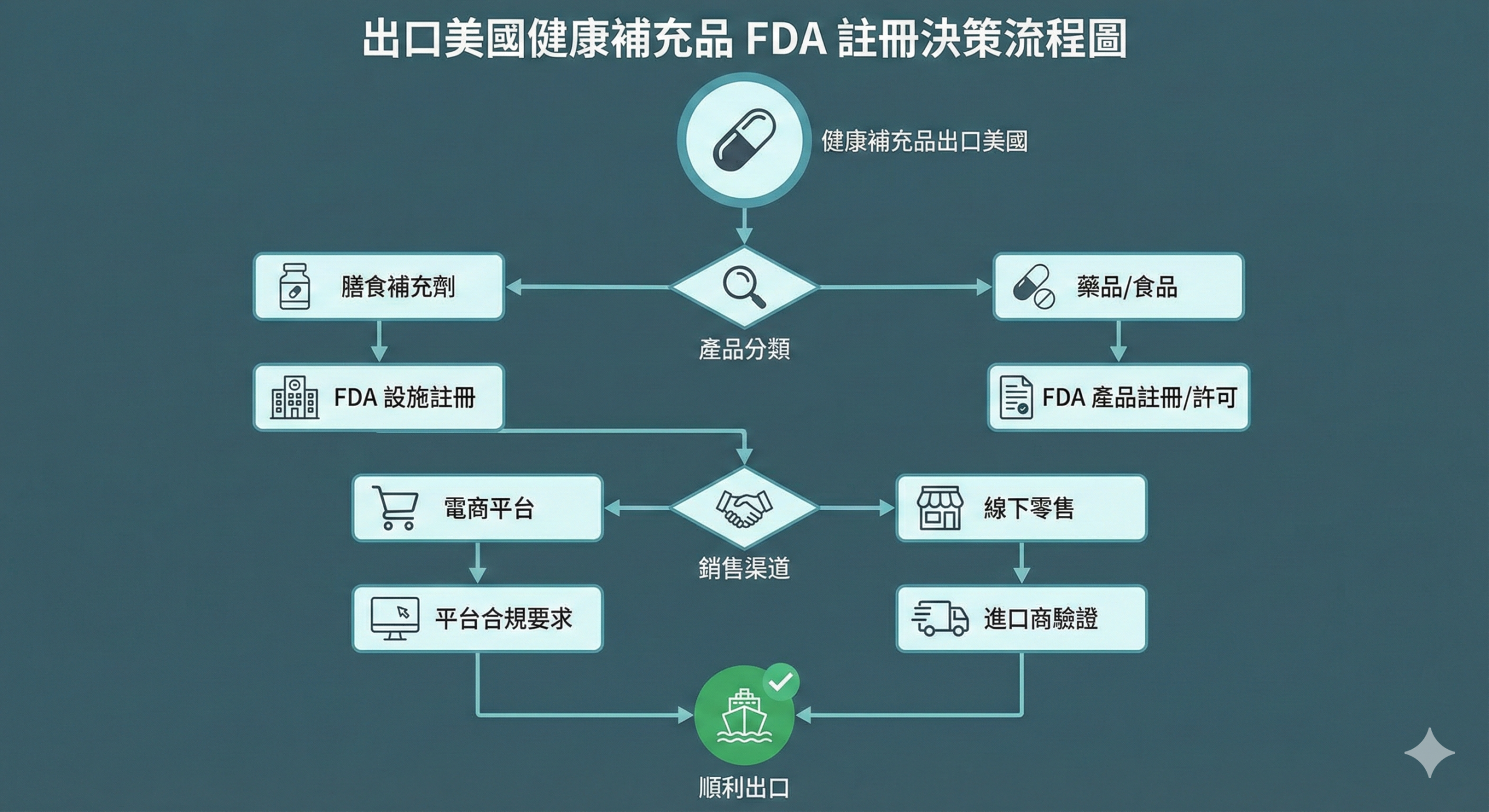
Whether a health food product requires FDA registration for export to the U.S. depends on whether the product is categorized as a "Dietary Supplement" or a "Conventional Food," as well as the requirements for the distribution channel. If the product meets the definition of Dietary Supplement and is sold through platforms such as Amazon, FDA registration of the food facility is usually required; if it is a Conventional Food or is imported only through private channels, it is determined on a case-by-case basis.
The logic of categorizing health food in the United States
The U.S. FDA categorizes "health food" into "Dietary Supplement" and "Conventional Food" based on the product form, ingredient labeling, and description of use.
Dietary supplements are usually in the form of capsules, tablets, powders, or liquids with "Supplement Facts" on the label, and contain vitamins, minerals, herbal extracts, or other nutrients. These products are required to comply with the Dietary Supplement Health and Education Act (DSHEA) and have a label that states, "This product has not been evaluated by the FDA and is not intended to diagnose, treat or prevent any disease".
General food products are labeled with "Nutrition Facts" for the primary purpose of consumption and include energy bars, functional beverages, or everyday food products with added nutrients. These products are subject to general food regulations and are not subject to specific regulations for dietary supplements.

(This chart illustrates the differences between dietary supplements and general food products in terms of product form, labeling, and use to assist sellers in determining product suitability.)
Applicable Contexts for FDA Food Facility Registration
Under the Bioterrorism Act and the Food Safety Modernization Act (FSMA), any facility that manufactures, processes, packages, or stores food for consumption in the United States is required to register as a food facility with the FDA.
This requirement applies to all food categories, including dietary supplements and general food products. If a Taiwanese manufacturer or packager is exporting to the U.S. market and the product will enter the U.S. for distribution, the facility will need to complete FDA registration.
The registration process includes providing basic information such as facility name, address, contact information, product category, and designating a U.S. Agent to act as a liaison between FDA and the facility. Upon completion of registration, the facility will receive a Registration Number, which can be used for customs reporting and platform review.
Audit requirements for e-commerce platforms such as Amazon
Amazon and other U.S. e-commerce platforms have a proprietary review mechanism for food products, requiring sellers to provide an FDA food facility registration number as basic proof of product compliance.
Even if the product itself does not require additional certification (e.g., dietary supplements do not require premarket approval), the Platform requires sellers to provide a facility registration number to confirm that the product is from a legitimate facility registered with the FDA. Failure to do so may result in the product not being able to be listed or being removed from the Platform.
In addition, some platforms also require sellers to provide copies of product labels, ingredient lists or third-party test reports to comply with the platform's internal policies and consumer protection regulations.
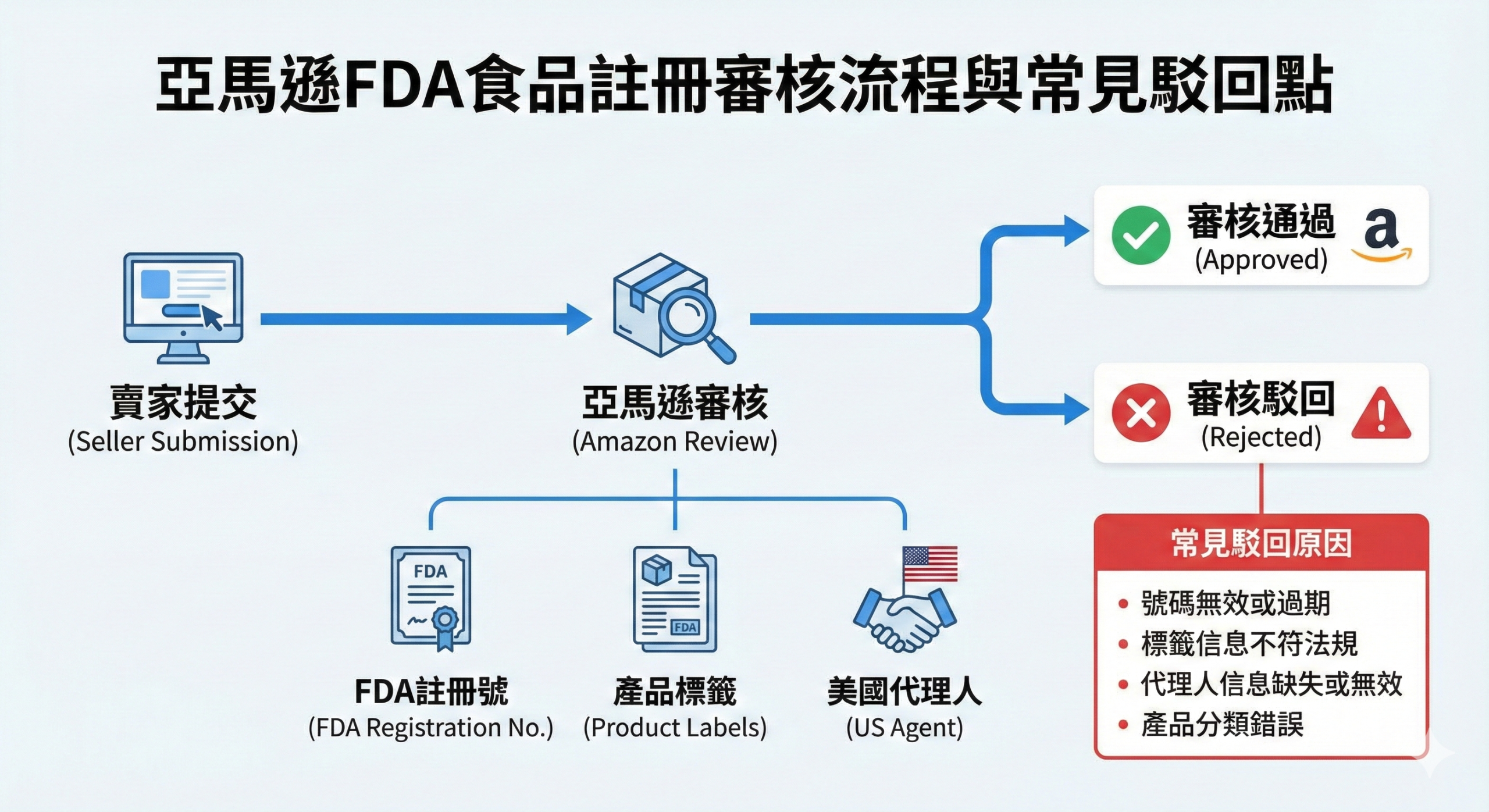
(This chart illustrates the Amazon platform's review requirements for FDA food facility registration, as well as common chokepoints and preparation points for sellers.)
Description of the formHealth Food Exports to the U.S. FDA Registration Judgment Table (Compilation of information: [Green Circle])
| Product Category | FDA registration required | Platform requirements | Remarks |
|---|---|---|---|
| Dietary Supplements (Sold by Amazon) | need | Registration number must be provided | Platform mandatory requirements |
| Dietary supplements (private import) | need | Subject to importer's requirements | Recommended to complete registration |
| General Foods (sold by Amazon) | need | Registration number must be provided | Platform mandatory requirements |
| General foodstuffs (private import) | need | Subject to importer's requirements | Recommended to complete registration |
textual criticism::
(This table organizes the applicable circumstances and platform requirements for FDA food facility registration under different product categories and sales channels to assist sellers in making quick judgments.)
Differences in judgment of different sales channels
If a product is sold on Amazon, eBay, or other U.S. e-commerce platforms, the platforms often mandate an FDA food facility registration number as a condition of listing. This is regardless of whether the product is a dietary supplement or a general food product.
If a product enters the U.S. market through a private importer or distributor, the need for FDA registration depends on the requirements of the importer and how the product is distributed. Some importers will require the supplier to provide a registration number to minimize the risk of Customs inspection, while others will only require compliance with product labeling and ingredient specifications.
Even if the products are not sold through the platform, it is still recommended that Taiwanese manufacturers or packagers take the initiative to complete the FDA food facility registration in order to establish the basis for product compliance and to reduce the risk of being detained by Customs or asked to make up the documents due to the lack of registration in the future.
[Application Notes
The content of this article is based on currently available official information, the actual application of the latest announcements and actual implementation of the competent authorities shall prevail.
[Official Supporting Documents
According to the FDA's public instructions, all facilities that manufacture, process, package, or store food for consumption in the United States are required to register their food facilities with the FDA in order to comply with the Bioterrorism Act and the Food Safety Modernization Act.
official source::
Food Facility Registration
https://www.fda.gov/food/registration-food-facilities-and-other-submissions/how-register-your-food-facility-fda
FAQ
Q1:Does health food export to the U.S. have to be FDA registered?
A1: If the product is a dietary supplement or general food product that will be exported to the U.S. market, the manufacturing or packaging facility will need to complete an FDA food facility registration. If the product is sold through a platform such as Amazon, the registration number is usually mandatory.
Q2: What is the difference between FDA registration of dietary supplements and food products?
A2: Both require registration of the food facility and the registration process is the same. The difference lies in the classification and labeling of the products, with dietary supplements being subject to DSHEA and general food products being subject to general food regulations.
Q3: Do I still need to register with FDA if I only sell through a private importer?
A3: According to the regulations, registration of facilities still needs to be completed, but in practice, it depends on the requirements of importers. It is recommended to take the initiative to complete the registration to minimize the risk of customs inspection.
Q4: How long does it take to register an FDA food facility?
A4: The online registration process takes about 1-2 hours to complete and the registration number takes effect immediately. Additional time will be required if you need to appoint a U.S. agent or prepare related documents.
Q5: Do I need to update my registration regularly?
A5: Facility registrations are subject to biennial renewal and changes in facility information must be notified to FDA within 60 days.
If you have not yet developed an overall understanding of FDA's food registration system, it is recommended that you start with a complete overview to help you later determine the different contexts and implementation differences.
👉 FDA Food Registration Guide|Taiwan Food Exports to U.S.A. Must Read (2026 Update)
For further information on the FDA food registration application process and required documents, please refer to the following pages to clarify practical points of judgment.
👉 FDA Food Registration Process and Documentation|Taiwan Application Highlights (2026)
If you are further concerned about the review requirements for FDA food registrations on the Amazon platform, you may wish to refer to the following help pages to clarify practical points of judgment.
Before planning, it is recommended to establish a proper understanding of the overall system and logic of judgment, and then conduct an assessment based on your own conditions, which will help reduce the uncertainty of subsequent decision-making and implementation.
For a more systematic understanding of this topic, please refer to the full description organized by [Green Circle].
[Description
This article is intended to be informative and is intended to assist in the understanding of the system and the logic of judgment, and does not constitute legal, regulatory or operational advice.